A recent announcement from the Centers for Disease Control and Prevention warrants a closer look since it may explain some of the issues that appeared during the 2021 influenza season.
Here is a Lab Alert from the CDC dated July 21, 2021:
The CDC is pre-warning clinic laboratories and testing sites that, effective January 1, 2022, the CDC is withdrawing its request to the Food and Drug Administration for the Emergency Use Authorization of its CDC 2019-Novel Coronavirus Real-Time RT-PCR Diagnostic Panel which was originally approved for use on February 4, 2020. Test sites are advised to use one of the other 250 RT-PCR COVID-19 authorized molecular diagnostic tools as shown on this screen capture (which shows only a few of the approved tests) with the soon-to-be withdrawn CDC product highlighted:
Here is the key phrase of the CDC's Lab Alert:
"CDC encourages laboratories to consider adoption of a multiplexed method that can facilitate detection and differentiation of SARS-CoV-2 and influenza viruses. Such assays can facilitate continued testing for both influenza and SARS-CoV-2 and can save both time and resources as we head into influenza season."
It would appear that the CDC's own RT-PCR diagnostic equipment is not capable of differentiating between influenza and COVID-19 and, as such, is being withdrawn from service before the 2022 influenza season begins.
This issue with the RT-PCR testing equipment may go a long way to explaining why there were very few positive tests for influenza over the 2021 season as shown here:
...when compared to the 2019 and 2020 influenza seasons as shown here (note the dramatic change in the y-axis scale):
This begs two questions since the entire narrative of the COVID-19 pandemic was built on the foundation of the RT-PCR tests:
1.) How many of the tests that were positive for the SARS-CoV-2 virus were actually detecting the virus that causes seasonal influenza during the 2021 flu season?
2.) How many of the other 250 RT-PCR COVID-19 molecular diagnostic tools that have been in use throughout the pandemic have the same issue of differentiating between the two viruses?
I'm guessing that we'll never know.
Addendum July 29, 2021
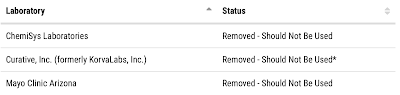
The Food and Drug Administration recently released information regarding RT-PCR tests that should no longer be used and/or distributed for COVID-19. Here are the most recent tests that have been withdrawn from use:
In addition to these 22 tests which were removed from service on the July 6 and July 15, 2021 updates, the FDA announced the removal of an additional 267 tests on the July 2, 2021 update.
Here's what the FDA had to say about the removal of RT-PCR tests:
"The FDA had previously included them on the website notification list of commercial manufacturers distributing serology test kits under that policy, but they have now been removed from that notification list and placed on the list below. As noted in the guidance, if an EUA request is not submitted by a commercial manufacturer of a serology test within a reasonable period of time, or if significant problems are identified with such a test that cannot be or have not been addressed in a timely manner, the FDA intends to remove the manufacturer and test from the notification list."
Tests may also be removed voluntarily by the manufacturer.






This conspiracy-driven “narrative” is a gross distortion of fact-based reality.
ReplyDeleteThe CDC PCR test is not perfect but was specific for Covid. There are just better tests now and one of the advantages is the practical possibility to test for more than one virus per specimen. Those newer tests, even if used simultaneously to test for two or more viruses, can reliably differentiate them (it’s not that hard because the viruses are very different).
https://pubmed.ncbi.nlm.nih.gov/34152951/
Covid case rates have been a very strong indicator of oncoming Covid deaths in the US, with a relative decoupling recently essentially due to the efficacy of vaccines. At least that's what the facts are strongly suggesting.
https://ourworldindata.org/grapher/daily-covid-cases-deaths?yScale=log&country=~USA
I am unaware where you get this crap from but it is 100% bullshit! And the RT-PCR test WAS NEVER SPECIFIC to "covid" or any other virus!
DeleteHow can there be a "covid test" for something that has never been isolated, purified or identified?! Until this happens, no amount of their fake tests will be worth a plugged nickle!!
This 'crap' comes from built knowledge over a long period with theoretical and practical applications. PCR test technology has been around for a while now, has no ties to any political or ideological tribe and it's possible to design a PCR test specific for a virus genome, including the coronavirus. It's kind of basic, if you know the basics of this test.
DeleteYou appear to regurgitate what's circulating in various echo-chambers. Is there a test for that? Yes, it's called fact-based objective analysis. :)
Isn't it fascinating that those who test positive (typically more than 95% of new cases, hospitalized, deaths) for the coronavirus these days belong to the unvaccinated group?
It's OK to believe that vaccines are not effective or dangerous but believing that this is a rational conclusion is a dangerous proposition, for the individuals, the community and the entire nation.