While you may or may not be aware of how the pharmaceutical industry funds the government bodies that regulate its activities and how there is a revolving door between the regulatory bodies and Big Pharma, a recent investigation by Maryanne Demasi which appeared on the British Medical Journals website certainly provides us with a very clear summary of the issue.
In the externally peer-reviewed article entitled "From FDA to MHRA: are drug regulators for hire?" the author's analysis opens by noting that both patients and physicians expect that government drug regulating bodies are independent from drug companies, allowing them provide an unbiased assessment of medical products before they reach the market. This lack of bias is the only thing standing in the way of approving medical products that are not safe for consumption or use.
Let's look at Demasi's analysis keeping in mind the following:
EMA - European Medicines Agency
FDA - United States Food and Drug Agency
MHRA - United Kingdom's Medicines and Healthcare Products Regulatory Agency
TGA - Australia's Therapeutic Goods Administration
HC - Health Canada
PDMA - Japan's Pharmaceuticals and Medical Devices Agency
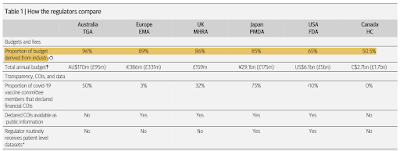
Here is a table summarizing the author's findings with CIO meaning Conflict of Interest:
It may surprise you to know that some of these government oversight bodies do not undertake an independent assessment of individual patient data, instead, they rely on an aggregation of data that is supplied to them by the company sponsoring the drug or device. In fact, only the FDA and PDMA obtain patient-level data which, of course, are not provided to the public. In a recent legal case, a group of professors and researchers called for the FDA to release all of the data that it used to grant Emergency Use Authorization for the COVID-19 vaccines. In response, the FDA claimed that the burden of supplying the data was too great and stated that it would release only 500 appropriately redacted pages per month, a process that would have taken 75 years to complete. Fortunately, a U.S. Federal Court judge ruled this unacceptable and ordered the FDA to release all of the data within 8 months.
Overall, the proportion of decisions to approve new medicines is shockingly high particularly given that many drugs are later recalled for significant safety issues. The proportion of decisions to approve new drugs is 98.5 percent in the United Kingdom, 98 percent in the United States, 94 percent in Australia, 88 percent in Europe and 83 percent in Canada (no disclosure from Japan). Interestingly given the rapidity with which the COVID-19 vaccines were released to the public, it shouldn't surprise anyone to find that all major regulators offer expedited pathways for the approval of new drugs; of new drugs approved in 2020, 68 percent in the United States, 50 percent in Europe, 36 percent in the United Kingdom, 26 percent in Japan, 20 percent in Australia and 16 percent in Canada were expedited. This means that the lengthy and thorough multi-phase trials of the past are becoming less and less common.
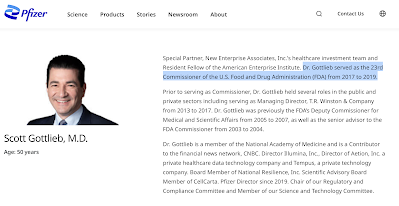
In the opening to this posting, I also noted the revolving door between the regulatory bodies and Big Pharma. In fact, according to the author, nine out of ten of the FDA's past commissioners between 2006 and 2019 found themselves in roles that were linked with pharmaceutical companies after departing from the world's most influential regulator. Here's a prime example of the problem which is particularly pertinent given the company's COVID-19 vaccine which received the EUA designation from the FDA:
It is very clear that these government regulatory bodies which rely on the very industry that they are designed to regulate for their funding and for the future employment of their employees, management and executive class is the very definition of a conflict of interest. and yet, they are in control of our safety when it comes to pharmaceutical products.





No comments:
Post a Comment